Stratification according to autoantibody status in systemic sclerosis reveals distinct molecular signatures
La stratification des patients sclérodermiques selon leur statut auto-anticorps révèle des signatures moléculaires distinctes.
Rouvière B., Le Dantec C., Bettacchioli E., Beretta L., Foulquier N., Cao C., Jamin C., Pers J.-O., Kerick M., Martin J., on behalf of PRECISESADS Clinical Consortium, on behalf of PRECISESADS Metabolomic Study Group, Alarcón-Riquelme M. E., de Moreuil C., Cornec D., Hillion S.
Ann Rheum Dis 2024, doi: 10.1136/ard-2024-225925
Background:
Despite advances in understanding in Systemic sclerosis (SSc) pathophysiology, current treatments focus only on managing organ complications. The Leroy classification (1988) divided SSc patients into two groups: diffuse and localized forms, based on skin involvement. However, recent findings by Elhai et al.1 suggest that autoantibodies are better prognostic markers than cutaneous subtype classification. The biological characteristics of different serotypes remain poorly understood.
Methods:
This study analyzed transcriptomic, metabolomic, cytometric, cytokine, and serological data from the PRECISESADS cohort. This European multicenter study included 955 patients with systemic autoimmune diseases to better characterize them through biological data.
Results:
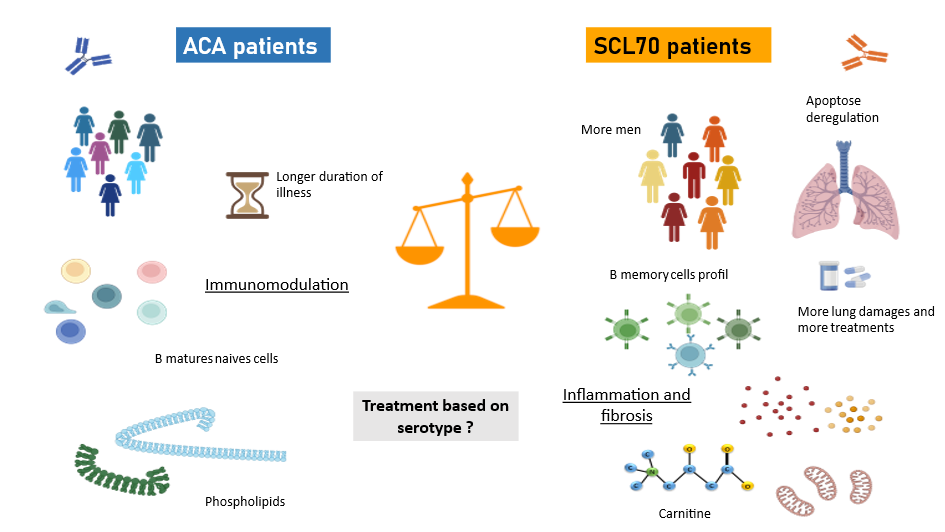
Among 244 patients with known serological status, 115 were anti-centromere antibody-positive (ACA), and 91 were anti-SCL70 (SCL70). SCL70 patients were younger and had higher skin, lung, and joint involvement. Transcriptomic data showed a marked neutrophil signature in SCL70 patients, along with decreased B-lymphocyte gene expression, except for memory B-cell genes, which were overexpressed. Unmedicated SCL70 (36) patients exhibited upregulation of genes involved in cell proliferation, epithelial and muscle differentiation, extracellular matrix organization, fibrosis, and apoptosis. Memory B-cell signatures were still present, with increased IGHG1, IGLC1, and IGHG4 expression. ACA untreated patients (91) showed a predominant skin development, tissue remodeling, and immunomodulation signature. Flow cytometry revealed increased neutrophil frequency and decreased T and B lymphocytes in SCL70 patients, with a shift from immature to memory B cells. Cytokine analysis showed elevated GDF-15, CCL17/TARC, and IL-6 levels in SCL70 patients, correlating with fibrosis. Metabolomics in 29 patients revealed increased oxidized fatty acids in SCL70, linked to inflammation, while ACA patients had elevated phosphatidylcholines and phosphatidylethanolamines, known to protect against lung fibrosis.
Conclusion:
This study confirms that SSc is a heterogeneous disease with distinct clinical, cytometric, and molecular signatures based on serological status. SCL70 patients exhibit a pro-inflammatory, pro-fibrotic signature, while ACA patients display an immunomodulatory and homeostatic profile. These findings could lead to a new patient classification and influence future therapeutic approaches.
Keywords: Systemic Sclerosis, Autoantibodies, Neutrophils, Fibrosis, Inflammation
Lien associé pubmed: https://pubmed.ncbi.nlm.nih.gov/39438128/
1.Elhai, M., Sritharan, N., Boubaya, M., Balbir-Gurman, A., Siegert, E., Hachulla, E., … & Kötter, I. (2022). Stratification in systemic sclerosis according to autoantibody status versus skin involvement: a study of the prospective EUSTAR cohort. The Lancet Rheumatology, 4(11), e785-e794.
